|
Question 1: Name the atom which has one electron, one proton and no neutron.Osteosarcoma (OST) is the most common bone tumor in children and adolescents with a second peak of incidence in elderly adults usually diagnosed as secondary tumors in Paget's disease or irradiated bone. Regardless of seeing the early atomic models were inaccurate and failed to explain certain experimental results, they were the base for future developments in the world of quantum mechanics. Rutherford also did not describe the arrangement of electrons in the orbit as one of the other drawbacks of his model.So Rutherford model has created a high contradiction with Maxwell’s theory and Rutherford later could not explain the stability of an atom. As per the Rutherford model, calculations have shown that an electron would collapse in the nucleus in less than 10 -8 seconds. 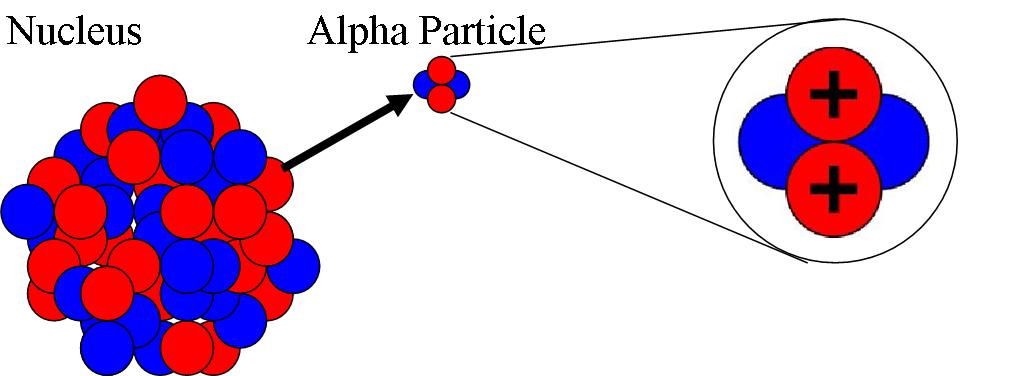
Ultimately electrons would collapse inside the nucleus. The radiation would carry energy from the motion which led to the shrinking of orbit. but Rutherford model says that the electrons revolve around the nucleus in fixed paths called orbits.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
